Kaist
Korean

Students News
-

A New Strategy for the Optimal Electroreduction of..
-Researchers suggest that modulation of local CO2 concentration improves the selectivity, conversion rate, and electrode stability, and shed a new light on the electrochemical CO2 reduction technology for controlling emissions at a low cost.- < (Clockwise from back left) PhD Candidate Hakhyeon Song, Professor Jihun Oh, Dr. Ying Chuan Tan, and M.S. Candidate Kelvin Berm Lee > A KAIST research team presented three novel approaches for modulating local carbon dioxide (CO2) concentration in gas-diffusion electrode (GDE)-based flow electrolyzers. Their study also empirically demonstrated that providing a moderate local CO2 concentration is effective in promoting Carbon–Carbon (C–C) coupling reactions toward the production of multi-carbon molecules. This work, featured in the May 20th issue of Joule, serves as a rational guide to tune CO2 mass transport for the optimal production of valuable multi-carbon products. Amid global efforts to reduce and recycle anthropogenic CO2 emissions, CO2 electrolysis holds great promise for converting CO2 into useful chemicals that were traditionally derived from fossil fuels. Many researches have been attempting to improve the selectivity of CO2 for commercially and industrially high-value multi-carbon products such as ethylene, ethanol, and 1-propanol, due to their high energy density and large market size. In order to achieve the highly-selective conversion of CO2 into valuable multi-carbon products, past studies have focused on the design of catalysts and the tuning of local environment related to pH, cations, and molecular additives. Conventional CO2 electrolytic systems relied heavily on an alkaline electrolyte that is often consumed in large quantities when reacting with CO2, and thus led to an increase in the operational costs. Moreover, the life span of a catalyst electrode was short, due to its inherent chemical reactivity. In their recent study, a group of KAIST researchers led by Professor Jihun Oh from the Department of Materials Science and Engineering reported that the local CO2 concentration has been an overlooked factor that largely affects the selectivity toward multi-carbon products. Professor Oh and his researchers Dr. Ying Chuan Tan, Hakhyeon Song, and Kelvin Berm Lee proposed that there is an intimate relation between local CO2 and multi-carbon product selectivity during electrochemical CO2 reduction reactions. The team employed the mass-transport modeling of a GDE-based flow electrolyzer that utilizes copper oxide (Cu2O) nanoparticles as model catalysts. They then identified and applied three approaches to modulate the local CO2 concentration within a GDE-based electrolytic system, including 1) controlling the catalyst layer structure, 2) CO2 feed concentration, and 3) feed flow rate. Contrary to common intuition, the study showed that providing a maximum CO2 transport leads to suboptimal multi-carbon product faradaic efficiency. Instead, by restricting and providing a moderate local CO2 concentration, C–C coupling can be significantly enhanced. The researchers demonstrated experimentally that the selectivity rate increased from 25.4% to 61.9%, and from 5.9% to 22.6% for the CO2 conversion rate. When a cheap milder near-neutral electrolyte was used, the stability of the CO2 electrolytic system improved to a great extent, allowing over 10 hours of steady selective production of multi-carbon products. Dr. Tan, the lead author of the paper, said, “Our research clearly revealed that the optimization of the local CO2 concentration is the key to maximizing the efficiency of converting CO2 into high-value multi-carbon products.” Professor Oh added, “This finding is expected to deliver new insights to the research community that variables affecting local CO2 concentration are also influential factors in the electrochemical CO2 reduction reaction performance. My colleagues and I hope that our study becomes a cornerstone for related technologies and their industrial applications.” This work was supported by the Korean Ministry of Science and ICT (MSIT) Creative Materials Discovery Program. < Figure. Three strategies employed in this study to modulate local CO2 concentration in a catalyst layer (top) and the relationship between local CO2 concentration and the selectivity for multi-carbon products (bottom). Note that maximum selectivity is achieved at a moderate local CO2 concentration. > Publication: Tan, Y. C et al. (2020) ‘Modulating Local CO2 Concentration as a General Strategy for Enhancing C−C Coupling in CO2 Electroreduction’, Joule, Vol. 4, Issue 5, pp. 1104-1120. Available online at https://doi.org/10.1016/j.joule.2020.03.013 Profile: Jihun Oh, PhD Associate Professor jihun.oh@kaist.ac.kr http://les.kaist.ac.kr/ Laboratory for Energy and Sustainability (LE&S) Department of Materials Science and Engineering (MSE) Korea Advanced Institute of Science and Technology (KAIST) https://www.kaist.ac.kr Daejeon 34141, Republic of Korea Profile: Ying Chuan Tan, PhD tanyc@kaist.ac.kr LE&S, MSE, KAIST Profile: Hakhyeon Song, PhD Candidate hyeon0401@kaist.ac.kr LE&S, MSE, KAIST Profile: Kelvin Berm Lee, M.S. Candidate kbl9105@kaist.ac.kr LE&S, MSE, KAIST (END)
-

Energy Storage Using Oxygen to Boost Battery Perfo..
Researchers have presented a novel electrode material for advanced energy storage device that is directly charged with oxygen from the air. Professor Jeung Ku Kang’s team synthesized and preserved the sub-nanometric particles of atomic cluster sizes at high mass loadings within metal-organic frameworks (MOF) by controlling the behavior of reactants at the molecular level. This new strategy ensures high performance for lithium-oxygen batteries, acclaimed as a next-generation energy storage technology and widely used in electric vehicles. Lithium-oxygen batteries in principle can generate ten times higher energy densities than conventional lithium-ion batteries, but they suffer from very poor cyclability. One of the methods to improve cycle stability is to reduce the overpotential of electrocatalysts in cathode electrodes. When the size of an electrocatalyst material is reduced to the atomic level, the increased surface energy leads to increased activity while significantly accelerating the material’s agglomeration. As a solution to this challenge, Professor Kang from the Department of Materials Science and Engineering aimed to maintain the improved activity by stabilizing atomic-scale sized electrocatalysts into the sub-nanometric spaces. This is a novel strategy for simultaneously producing and stabilizing atomic-level electrocatalysts within metal-organic frameworks (MOFs). Metal-organic frameworks continuously assemble metal ions and organic linkers. The team controlled hydrogen affinities between water molecules to separate them and transfer the isolated water molecules one by one through the sub-nanometric pores of MOFs. The transferred water molecules reacted with cobalt ions to form di-nuclear cobalt hydroxide under precisely controlled synthetic conditions, then the atomic-level cobalt hydroxide is stabilized inside the sub-nanometric pores. The di-nuclear cobalt hydroxide that is stabilized in the sub-nanometric pores of metal-organic frameworks (MOFs) reduced the overpotential by 63.9% and showed ten-fold improvements in the life cycle. Professor Kang said, “Simultaneously generating and stabilizing atomic-level electrocatalysts within MOFs can diversify materials according to numerous combinations of metal and organic linkers. It can expand not only the development of electrocatalysts, but also various research fields such as photocatalysts, medicine, the environment, and petrochemicals.” This study was reported in Advanced Science (Title: Autogenous Production and Stabilization of Highly Loaded Sub-Nanometric Particles within Multishell Hollow Metal-Organic Frameworks and Their Utilization for High Performance in Li-O2 Batteries). This research was mainly supported by the Global Frontier R&D Program of the Ministry of Science, ICT & Planning (Grant No. 2013M3A6B1078884) funded by the Ministry of Science, ICT & Future Planning, and the National Research Foundation of Korea (Grant No. 2019M3E6A1104196). < Figure: Schematic of the formation process of EG–water complexes and illustration of the penetration process of an isolated water molecule. > Profile: Professor Jeung Ku Kang jeungku@kaist.ac.kr http://nanosf.kaist.ac.kr/ Nano Materials Simulation and Fabrication Laboratory Department of Materials Science and Engineering KAIST
-

Universal Virus Detection Platform to Expedite Vir..
Reactive polymer-based tester pre-screens dsRNAs of a wide range of viruses without their genome sequences < From left: Professor Yoosik Kim, Professor Sheng Li, PhD candidate Sura Kim, PhD candidate Jayoung Ku. > The prompt, precise, and massive detection of a virus is the key to combat infectious diseases such as Covid-19. A new viral diagnostic strategy using reactive polymer-grafted, double-stranded RNAs will serve as a pre-screening tester for a wide range of viruses with enhanced sensitivity. Currently, the most widely using viral detection methodology is polymerase chain reaction (PCR) diagnosis, which amplifies and detects a piece of the viral genome. Prior knowledge of the relevant primer nucleic acids of the virus is quintessential for this test. The detection platform developed by KAIST researchers identifies viral activities without amplifying specific nucleic acid targets. The research team, co-led by Professor Sheng Li and Professor Yoosik Kim from the Department of Chemical and Biomolecular Engineering, constructed a universal virus detection platform by utilizing the distinct features of the PPFPA-grafted surface and double-stranded RNAs. The key principle of this platform is utilizing the distinct feature of reactive polymer-grafted surfaces, which serve as a versatile platform for the immobilization of functional molecules. These activated surfaces can be used in a wide range of applications including separation, delivery, and detection. As long double-stranded RNAs are common byproducts of viral transcription and replication, these PPFPA-grafted surfaces can detect the presence of different kinds of viruses without prior knowledge of their genomic sequences. “We employed the PPFPA-grafted silicon surface to develop a universal virus detection platform by immobilizing antibodies that recognize double-stranded RNAs,” said Professor Kim. To increase detection sensitivity, the research team devised two-step detection process analogues to sandwich enzyme-linked immunosorbent assay where the bound double-stranded RNAs are then visualized using fluorophore-tagged antibodies that also recognize the RNAs’ double-stranded secondary structure. By utilizing the developed platform, long double-stranded RNAs can be detected and visualized from an RNA mixture as well as from total cell lysates, which contain a mixture of various abundant contaminants such as DNAs and proteins. The research team successfully detected elevated levels of hepatitis C and A viruses with this tool. “This new technology allows us to take on virus detection from a new perspective. By targeting a common biomarker, viral double-stranded RNAs, we can develop a pre-screening platform that can quickly differentiate infected populations from non-infected ones,” said Professor Li. “This detection platform provides new perspectives for diagnosing infectious diseases. This will provide fast and accurate diagnoses for an infected population and prevent the influx of massive outbreaks,” said Professor Kim. This work is featured in Biomacromolecules. This work was supported by the Agency for Defense Development (Grant UD170039ID), the Ministry of Science and ICT (NRF-2017R1D1A1B03034660, NRF-2019R1C1C1006672), and the KAIST Future Systems Healthcare Project from the Ministry of Science and ICT (KAISTHEALTHCARE42). < Figure: Schematics of the reactive polymer-coated surface for dsRNA capture and detection. > Profile: -Professor Yoosik Kim Department of Chemical and Biomolecular Engineering https://qcbio.kaist.ac.kr KAIST -Professor Sheng Li Department of Chemical and Biomolecular Engineering https://bcpolymer.kaist.ac.kr KAIST Publication: Ku et al., 2020. Reactive Polymer Targeting dsRNA as Universal Virus Detection Platform with Enhanced Sensitivity. Biomacromolecules (https://doi.org/10.1021/acs.biomac.0c00379).
-
New Nanoparticle Drug Combination For Atherosclero..
< Professor Ji-Ho Park (left) and Dr. Heegon Kim (right) > Physicochemical cargo-switching nanoparticles (CSNP) designed by KAIST can help significantly reduce cholesterol and macrophage foam cells in arteries, which are the two main triggers for atherosclerotic plaque and inflammation. The CSNP-based combination drug delivery therapy was proved to exert cholesterol-lowering, anti-inflammatory, and anti-proliferative functions of two common medications for treating and preventing atherosclerosis that are cyclodextrin and statin. Professor Ji-Ho Park and Dr. Heegon Kim from KAIST’s Department of Bio and Brain Engineering said their study has shown great potential for future applications with reduced side effects. Atherosclerosis is a chronic inflammatory vascular disease that is characterized by the accumulation of cholesterol and cholesterol-loaded macrophage foam cells in the intima. When this atherosclerotic plaque clogs and narrows the artery walls, they restrict blood flow and cause various cardiovascular conditions such as heart attacks and strokes. Heart attacks and strokes are the world’s first and fifth causes of death respectively. Oral statin administration has been used in clinics as a standard care for atherosclerosis, which is prescribed to lower blood cholesterol and inhibit its accumulation within the plaque. Although statins can effectively prevent the progression of plaque growth, they have only shown modest efficacy in eliminating the already-established plaque. Therefore, patients are required to take statin drugs for the rest of their lives and will always carry the risk of plaque ruptures that can trigger a blood clot. To address these issues, Professor Park and Dr. Kim exploited another antiatherogenic agent called cyclodextrin. In their paper published in the Journal of Controlled Release on March 10, Professor Park and Dr. Kim reported that the polymeric formulation of cyclodextrin with a diameter of approximately 10 nanometers(nm) can accumulate within the atherosclerotic plaque 14 times more and effectively reduce the plaque even at lower doses, compared to cyclodextrin in a non-polymer structure. Moreover, although cyclodextrin is known to have a cytotoxic effect on hair cells in the cochlea, which can lead to hearing loss, cyclodextrin polymers developed by Professor Park’s research group exhibited a varying biodistribution profile and did not have this side effect. In the follow-up study reported in ACS Nano on April 28, the researchers exploited both cyclodextrin and statin and form the cyclodextrin-statin self-assembly drug complex, based on previous findings that each drug can exert local anti-atherosclerosis effect within the plaque. The complex formation processes were optimized to obtain homogeneous and stable nanoparticles with a diameter of about 100 nm for systematic injection. The therapeutic synergy of cyclodextrin and statin could reportedly enhance plaque-targeted drug delivery and anti-inflammation. Cyclodextrin led to the regression of cholesterol in the established plaque, and the statins were shown to inhibit the proliferation of macrophage foam cells. The study suggested that combination therapy is required to resolve the complex inflammatory cholesterol-rich microenvironment within the plaque. Professor Park said, “While nanomedicine has been mainly developed for the treatment of cancers, our studies show that nanomedicine can also play a significant role in treating and preventing atherosclerosis, which causes various cardiovascular diseases that are the leading causes of death worldwide.” This work was supported by KAIST and the National Research Foundation (NRF) of Korea. < Figure. Antiatherogenic effect of CSNP. > Publications: 1. Heegon Kim, Junhee Han, and Ji-Ho Park. (2020) ‘Cyclodextrin polymer improves atherosclerosis therapy and reduces ototoxicity’ Journal of Controlled Release. Volume 319. Page 77-86. Available online at https://doi.org/10.1016/j.jconrel.2019.12.021 2. Kim, H., et al. (2020) ‘Affinity-Driven Design of Cargo-Switching Nanoparticles to Leverage a Cholesterol-Rich Microenvironment for Atherosclerosis Therapy’ ACS Nano. Available online at https://doi.org/10.1021/acsnano.9b08216 Profile: Ji-Ho Park, Ph.D. Associate Professor jihopark@kaist.ac.kr http://openwetware.org/wiki/Park_Lab Biomaterials Engineering Laboratory (BEL) Department of Bio and Brain Engineering (BIOENG) Korea Advanced Institute of Science and Technology (KAIST) https://www.kaist.ac.kr Daejeon 34141, Korea Profile: Heegon Kim, Ph.D. Postdoctoral Researcher heegon@kaist.ac.kr BEL, BIOENG, KAIST (END)
-

A Deep-Learned E-Skin Decodes Complex Human Motion
A deep-learning powered single-strained electronic skin sensor can capture human motion from a distance. The single strain sensor placed on the wrist decodes complex five-finger motions in real time with a virtual 3D hand that mirrors the original motions. The deep neural network boosted by rapid situation learning (RSL) ensures stable operation regardless of its position on the surface of the skin. Conventional approaches require many sensor networks that cover the entire curvilinear surfaces of the target area. Unlike conventional wafer-based fabrication, this laser fabrication provides a new sensing paradigm for motion tracking. The research team, led by Professor Sungho Jo from the School of Computing, collaborated with Professor Seunghwan Ko from Seoul National University to design this new measuring system that extracts signals corresponding to multiple finger motions by generating cracks in metal nanoparticle films using laser technology. The sensor patch was then attached to a user’s wrist to detect the movement of the fingers. The concept of this research started from the idea that pinpointing a single area would be more efficient for identifying movements than affixing sensors to every joint and muscle. To make this targeting strategy work, it needs to accurately capture the signals from different areas at the point where they all converge, and then decoupling the information entangled in the converged signals. To maximize users’ usability and mobility, the research team used a single-channeled sensor to generate the signals corresponding to complex hand motions. The rapid situation learning (RSL) system collects data from arbitrary parts on the wrist and automatically trains the model in a real-time demonstration with a virtual 3D hand that mirrors the original motions. To enhance the sensitivity of the sensor, researchers used laser-induced nanoscale cracking. This sensory system can track the motion of the entire body with a small sensory network and facilitate the indirect remote measurement of human motions, which is applicable for wearable VR/AR systems. The research team said they focused on two tasks while developing the sensor. First, they analyzed the sensor signal patterns into a latent space encapsulating temporal sensor behavior and then they mapped the latent vectors to finger motion metric spaces. Professor Jo said, “Our system is expandable to other body parts. We already confirmed that the sensor is also capable of extracting gait motions from a pelvis. This technology is expected to provide a turning point in health-monitoring, motion tracking, and soft robotics.” This study was featured in Nature Communications. < Figure 1: Deep Learned Sensor collecting epicentral motion > < Figure 2: RSL system based on transfer learning > Publication: Kim, K.K., Ha, I., Kim, M. et al. A deep-learned skin sensor decoding the epicentral human motions. Nature Communications 11, 2149 (2020). https://doi.org/10.1038/s41467-020-16040-y29 Link to download the full-text paper: https://www.nature.com/articles/s41467-020-16040-y.pdf Profile: Professor Sungho Jo Neuro-Machine Augmented Intelligence Lab http://nmail.kaist.ac.kr School of Computing College of Engineering KAIST
-

Visualization of Functional Components to Characte..
< Dr. Hongjun Kim (left) and Professor Seungbum Hong (right) > Researchers have developed a visualization method that will determine the distribution of components in battery electrodes using atomic force microscopy. The method provides insights into the optimal conditions of composite electrodes and takes us one step closer to being able to manufacture next-generation all-solid-state batteries. Lithium-ion batteries are widely used in smart devices and vehicles. However, their flammability makes them a safety concern, arising from potential leakage of liquid electrolytes. All-solid-state lithium ion batteries have emerged as an alternative because of their better safety and wider electrochemical stability. Despite their advantages, all-solid-state lithium ion batteries still have drawbacks such as limited ion conductivity, insufficient contact areas, and high interfacial resistance between the electrode and solid electrolyte. To solve these issues, studies have been conducted on composite electrodes in which lithium ion conducting additives are dispersed as a medium to provide ion conductive paths at the interface and increase the overall ionic conductivity. It is very important to identify the shape and distribution of the components used in active materials, ion conductors, binders, and conductive additives on a microscopic scale for significantly improving the battery operation performance. The developed method is able to distinguish regions of each component based on detected signal sensitivity, by using various modes of atomic force microscopy on a multiscale basis, including electrochemical strain microscopy and lateral force microscopy. For this research project, both conventional electrodes and composite electrodes were tested, and the results were compared. Individual regions were distinguished and nanoscale correlation between ion reactivity distribution and friction force distribution within a single region was determined to examine the effect of the distribution of binder on the electrochemical strain. The research team explored the electrochemical strain microscopy amplitude/phase and lateral force microscopy friction force dependence on the AC drive voltage and the tip loading force, and used their sensitivities as markers for each component in the composite anode. This method allows for direct multiscale observation of the composite electrode in ambient condition, distinguishing various components and measuring their properties simultaneously. Lead author Dr. Hongjun Kim said, “It is easy to prepare the test sample for observation while providing much higher spatial resolution and intensity resolution for detected signals.” He added, “The method also has the advantage of providing 3D surface morphology information for the observed specimens.” Professor Seungbum Hong from the Department of Material Sciences and Engineering said, “This analytical technique using atomic force microscopy will be useful for quantitatively understanding what role each component of a composite material plays in the final properties.” “Our method not only will suggest the new direction for next-generation all-solid-state battery design on a multiscale basis but also lay the groundwork for innovation in the manufacturing process of other electrochemical materials.” This study is published in ACS Applied Energy Materials and supported by the Big Science Research and Development Project under the Ministry of Science and ICT and the National Research Foundation of Korea, the Basic Research Project under the Wearable Platform Materials Technology Center, and KAIST Global Singularity Research Program for 2019 and 2020. < Figure. AFM images of (a, c) samples A and (b, d) B. (a, b) Topographic height images and (c, d) friction force images, respectively. All images were acquired with a tip loading force of 400 nN. Red lines are guides for the eye. > Profile: Seungbum Hong Professor seungbum@kaist.ac.kr http://mii.kaist.ac.kr/ Materials Imaging and Integration Laboratory Department of Material Sciences and Engineering KAIST
-

From Dark to Light in a Flash: Smart Film Lets Win..
< (Clockwise from back left) Professor Jung-Wuk Hong, Professor Seokwoo Jeon, Dr. Sang-Eon Lee, and PhD Candidate Donghwi Cho > Researchers have developed a new easy-to-use smart optical film technology that allows smart window devices to autonomously switch between transparent and opaque states in response to the surrounding light conditions. The proposed 3D hybrid nanocomposite film with a highly periodic network structure has empirically demonstrated its high speed and performance, enabling the smart window to quantify and self-regulate its high-contrast optical transmittance. As a proof of concept, a mobile-app-enabled smart window device for Internet of Things (IoT) applications has been realized using the proposed smart optical film with successful expansion to the 3-by-3-inch scale. This energy-efficient and cost-effective technology holds great promise for future use in various applications that require active optical transmission modulation. Flexible optical transmission modulation technologies for smart applications including privacy-protection windows, zero-energy buildings, and beam projection screens have been in the spotlight in recent years. Conventional technologies that used external stimuli such as electricity, heat, or light to modulate optical transmission had only limited applications due to their slow response speeds, unnecessary color switching, and low durability, stability, and safety. The optical transmission modulation contrast achieved by controlling the light scattering interfaces on non-periodic 2D surface structures that often have low optical density such as cracks, wrinkles, and pillars is also generally low. In addition, since the light scattering interfaces are exposed and not subject to any passivation, they can be vulnerable to external damage and may lose optical transmission modulation functions. Furthermore, in-plane scattering interfaces that randomly exist on the surface make large-area modulation with uniformity difficult. Inspired by these limitations, a KAIST research team led by Professor Seokwoo Jeon from the Department of Materials Science and Engineering and Professor Jung-Wuk Hong of the Civil and Environmental Engineering Department used proximity-field nanopatterning (PnP) technology that effectively produces highly periodic 3D hybrid nanostructures, and an atomic layer deposition (ALD) technique that allows the precise control of oxide deposition and the high-quality fabrication of semiconductor devices. The team then successfully produced a large-scale smart optical film with a size of 3 by 3 inches in which ultrathin alumina nanoshells are inserted between the elastomers in a periodic 3D nanonetwork. This “mechano-responsive” 3D hybrid nanocomposite film with a highly periodic network structure is the largest smart optical transmission modulation film that exists. The film has been shown to have state-of-the-art optical transmission modulation of up to 74% at visible wavelengths from 90% initial transmission to 16% in the scattering state under strain. Its durability and stability were proved by more than 10,000 tests of harsh mechanical deformation including stretching, releasing, bending, and being placed under high temperatures of up to 70°C. When this film was used, the transmittance of the smart window device was adjusted promptly and automatically within one second in response to the surrounding light conditions. Through these experiments, the underlying physics of optical scattering phenomena occurring in the heterogeneous interfaces were identified. Their findings were reported in the online edition of Advanced Science on April 26. KAIST Professor Jong-Hwa Shin’s group and Professor Young-Seok Shim at Silla University also collaborated on this project. Donghwi Cho, a PhD candidate in materials science and engineering at KAIST and co-lead author of the study, said, “Our smart optical film technology can better control high-contrast optical transmittance by relatively simple operating principles and with low energy consumption and costs.” “When this technology is applied by simply attaching the film to a conventional smart window glass surface without replacing the existing window system, fast switching and uniform tinting are possible while also securing durability, stability, and safety. In addition, its wide range of applications for stretchable or rollable devices such as wall-type displays for a beam projection screen will also fulfill aesthetic needs,” he added. This work was supported by the National Research Foundation of Korea (NRF), and the Korean Ministries of Science, ICT and Future Planning (MSIP), and Science and ICT (MSIT). < Figure 1. Design concept of and fabrication procedures for the 3D scatterer > < Figure 2. Mechanical and optical simulations of the 3D scatterer > < Figure 3. Demonstrations of the internet of things (IoT) applications: a self-regulating mechano-responsive smart window (MSW) device and a beam projection screen > Publication: Cho, D, et al. (2020) ‘High-Contrast Optical Modulation from Strain-Indicated Nanogaps at 3D Heterogeneous Interfaces’ Advanced Science, 1903708. Available online at https://doi.org/10.1002/advs.201903708 Profile: Seokwoo Jeon, PhD Professor jeon39@kaist.ac.kr https://fdml.kaist.ac.kr/ Flexible Device and Metamaterials Lab (FDML) Department of Materials Science and Engineering (MSE) Korea Advanced Institute of Science and Technology (KAIST) https://www.kaist.ac.kr Daejeon 34141, Korea Profile: Jung-Wuk Hong, PhD Associate Professor j.hong@kaist.ac.kr http://aaml.kaist.ac.kr Advanced Applied Mechanics Laboratory (AAML) Department of Civil and Environmental Engineering KAIST Profile: Donghwi Cho PhD Candidate roy0202@kaist.ac.kr FDML, MSE, KAIST Profile: Young-Seok Shim, PhD Assistant Professor ysshim@silla.ac.kr Division of Materials Science and Engineering Silla University https://www.silla.ac.kr Busan 46958, Korea (END)
-

Dr. Dong-Hyun Cho at KARI Receives the 16th Jeong ..
< From left: PhD candidate Yongtae Yun at KAIST, Dr. Dong-Hyun Cho at KARI, Seonju Yim at Kongju National University High School, and MS-PhD candidate Haun-Min Lee at Korea University > Dr. Dong-Hyun Cho, a senior researcher at the Korea Aerospace Research Institute (KARI), was honored as the recipient of the 16th Jeong Hun Cho Award. The award recognizes young scientists in the field of aerospace engineering. Dr. Cho earned his MS and PhD degrees from the KAIST Department of Aerospace Engineering in 2012, and served as a researcher at the Satellite Technology Research Center (SaTReC) at KAIST, before joining the Future Convergence Research Division at KARI. He won this year’s award and received 25 million KRW in prize money. Jeong Hun Cho, who was a PhD candidate in the Department of Aerospace Engineering at KAIST, passed away in a tragic lab accident in May 2003 and was awarded an honorary doctorate posthumously. His family endowed the award and scholarship in his memory. Since 2005, the scholarship has selected three young scholars every year who specialize in aerospace engineering from Cho’s alma maters of KAIST, Korea University, and Kongju National University High School. Dr. Dong-Hyun Cho was selected as this year’s awardee in recognition of his studies on the development and operation of KARISMA, a comprehensive software package for space debris collision risk management. Dr. Cho built a terrestrial testbed and produced a model for the development of a space debris elimination algorithm. He published six papers in SCI-level journals and wrote 35 symposium papers in the field of space development. He also applied or registered approximately 40 patents both in Korea and internationally. The Award Committee also selected three students as scholarship recipients: PhD candidate Yongtae Yun from the Department of Aerospace Engineering at KAIST received 4 million KRW, MS-PhD candidate Haun-Min Lee from the School of Mechanical Engineering at Korea University received 4 million KRW, and Seonju Yim from Kongju National University High School received 3 million KRW. (END)
-
Researchers Present a Microbial Strain Capable of ..
< From left:Ph.D.candidate Gi Bae Kim, Dr.Jung Ho Ahn, Ph.D.candiate Jong An Lee, and Distinguished Professor Sang Yup Lee > A research team led by Distinguished Professor Sang Yup Lee reported the production of a microbial strain capable of the massive production of succinic acid with the highest production efficiency to date. This strategy of integrating systems metabolic engineering with enzyme engineering will be useful for the production of industrially competitive bio-based chemicals. Their strategy was described in Nature Communications on April 23. The bio-based production of industrial chemicals from renewable non-food biomass has become increasingly important as a sustainable substitute for conventional petroleum-based production processes relying on fossil resources. Here, systems metabolic engineering, which is the key component for biorefinery technology, is utilized to effectively engineer the complex metabolic pathways of microorganisms to enable the efficient production of industrial chemicals. Succinic acid, a four-carbon dicarboxylic acid, is one of the most promising platform chemicals serving as a precursor for industrially important chemicals. Among microorganisms producing succinic acid, Mannheimia succiniciproducens has been proven to be one of the best strains for succinic acid production. The research team has developed a bio-based succinic acid production technology using the M. succiniciproducens strain isolated from the rumen of Korean cow for over 20 years and succeeded in developing a strain capable of producing succinic acid with the highest production efficiency. They carried out systems metabolic engineering to optimize the succinic acid production pathway of the M. succiniciproducens strain by determining the crystal structure of key enzymes important for succinic acid production and performing protein engineering to develop enzymes with better catalytic performance. As a result, 134 g per liter of succinic acid was produced from the fermentation of an engineered strain using glucose, glycerol, and carbon dioxide. They were able to achieve 21 g per liter per hour of succinic acid production, which is one of the key factors determining the economic feasibility of the overall production process. This is the world’s best succinic acid production efficiency reported to date. Previous production methods averaged 1~3 g per liter per hour. Distinguished professor Sang Yup Lee explained that his team’s work will significantly contribute to transforming the current petrochemical-based industry into an eco-friendly bio-based one. “Our research on the highly efficient bio-based production of succinic acid from renewable non-food resources and carbon dioxide has provided a basis for reducing our strong dependence on fossil resources, which is the main cause of the environmental crisis,” Professor Lee said. This work was supported by the Technology Development Program to Solve Climate Changes via Systems Metabolic Engineering for Biorefineries and the C1 Gas Refinery Program from the Ministry of Science and ICT through the National Research Foundation of Korea. < Figure: Protein engineering of key enzymes corresponding to succinic acid production. >
-

Highly Efficient and Stable Double Layer Solar Cel..
< (Front row from left) Professor Byungha Shin (KAIST), and PhD Candidate Daehan Kim (KAIST). (Back row from left) Professor Jin Young Kim (Seoul National University), Dr. Ik Jae Park (Seoul National University), and Professor Dong Hoe Kim (Sejong University). > Solar cells convert light into energy, but they can be inefficient and vulnerable to the environment, degrading with, ironically, too much light or other factors, including moisture and low temperature. An international research team has developed a new type of solar cell that can both withstand environmental hazards and is 26.7% more efficient in power conversion. They published their results on March 26 in Science. The researchers, led by Byungha Shin, a professor from the Department of Materials Science and Engineering at KAIST, focused on developing a new class of light-absorbing material, called a wide bandgap perovskite. The material has a highly effective crystal structure that can process the power needs, but it can become problematic when exposed to environmental hazards, such as moisture. Researchers have made some progress increasing the efficiency of solar cells based on perovskite, but the material has greater potential than what was previously achieved. To achieve better performance, Shin and his team built a double layer solar cell, called tandem, in which two or more light absorbers are stacked together to better utilize solar energy. To use perovskite in these tandem devices, the scientists modified the material’s optical property, which allows it to absorb a wider range of solar energy. Without the adjustment, the material is not as useful in achieving high performing tandem solar cells. The modification of the optical property of perovskite, however, comes with a penalty — the material becomes hugely vulnerable to the environment, in particular, to light. To counteract the wide bandgap perovskite’s delicate nature, the researchers engineered combinations of molecules composing a two-dimensional layer in the perovskite, stabilizing the solar cells. “We developed a high-quality wide bandgap perovskite material and, in combination with silicon solar cells, achieved world-class perovskite-silicon tandem cells,” Shin said. The development was only possible due to the engineering method, in which the mixing ratio of the molecules building the two-dimensional layer are carefully controlled. In this case, the perovskite material not only improved efficiency of the resulting solar cell but also gained durability, retaining 80% of its initial power conversion capability even after 1,000 hours of continuous illumination. This is the first time such a high efficiency has been achieved with a wide bandgap perovskite single layer alone, according to Shin. “Such high-efficiency wide bandgap perovskite is an essential technology for achieving ultra-high efficiency of perovskite-silicon tandem (double layer) solar cells,” Shin said. “The results also show the importance of bandgap matching of upper and lower cells in these tandem solar cells.” The researchers, having stabilized the wide bandgap perovskite material, are now focused on developing even more efficient tandem solar cells that are expected to have more than 30% of power conversion efficiency, something that no one has achieved yet, “Our ultimate goal is to develop ultra-high-efficiency tandem solar cells that contribute to the increase of shared solar energy among all energy sources,” Shin said. “We want to contribute to making the planet healthier.” This work was supported by the National Research Foundation of Korea, the Korea Institute of Energy Technology Evaluation and Planning, the Ministry of Trade Industry and Energy of Korea, and the U.S. Department of Energy. Other contributors include Daehan Kim, Jekyung Kim, Passarut Boonmongkolras, Seong Ryul Pae and Minkyu Kim, all of whom affiliated with the Department of Materials Science and Engineering at KAIST. Other authors include Byron W. Larson, Sean P. Dunfield, Chuanxiao Xiao, Jinhui Tong, Fei Zhang, Joseph J. Berry, Kai Zhu and Dong Hoe Kim, all of who are affiliated with the National Renewable Energy Laboratory in Colorado. Dunfield is also affiliated with the Materials Science and Engineering Program at the University of Colorado; Berry is also affiliated with the Department of Physics and the Renewable and Sustainable Energy Institute at the University of Colorado Boulder; and Kim is also affiliated with the Department of Nanotechnology and Advanced Materials Engineering at Sejong University. Hee Joon Jung and Vinayak Dravid of the Department of Materials Science and Engineering at Northwestern University; Ik Jae Park, Su Geun Ji and Jin Young Kim of the Department of Materials Science and Engineering at Seoul National University; and Seok Beom Kang of the Department of Nanotechnology and Advanced Materials Engineering of Sejong University also contributed. < High-resolution TEM study revealing atomic configuration of the 2D passivation layers. > < Structure and photovoltaic performance for the perovskite-Si tandem device. > Image credit: Professor Byungha Shin, KAIST Image usage restrictions: News organizations may use or redistribute this image, with proper attribution, as part of news coverage of this paper only. Publication: Kim et al. (2020) “Efficient, stable silicon tandem cells enabled by anion-engineered wide band gap perovskites”. Science. Available online at https://doi.org/10.1126/science.aba3433 Profile: Byungha Shin Professor byungha@kaist.ac.kr http://energymatlab.kaist.ac.kr/ Department of Materials Science and Engineering KAIST (END)
-

Ultrathin but Fully Packaged High-Resolution Camer..
- Biologically inspired ultrathin arrayed camera captures super-resolution images. - < Ph.D. Candidate Kisoo Kim and Professor Ki-Hun Jeong > The unique structures of biological vision systems in nature inspired scientists to design ultracompact imaging systems. A research group led by Professor Ki-Hun Jeong have made an ultracompact camera that captures high-contrast and high-resolution images. Fully packaged with micro-optical elements such as inverted micro-lenses, multilayered pinhole arrays, and gap spacers on the image sensor, the camera boasts a total track length of 740 μm and a field of view of 73°. Inspired by the eye structures of the paper wasp species Xenos peckii, the research team completely suppressed optical noise between micro-lenses while reducing camera thickness. The camera has successfully demonstrated high-contrast clear array images acquired from tiny micro lenses. To further enhance the image quality of the captured image, the team combined the arrayed images into one image through super-resolution imaging. An insect’s compound eye has superior visual characteristics, such as a wide viewing angle, high motion sensitivity, and a large depth of field while maintaining a small volume of visual structure with a small focal length. Among them, the eyes of Xenos peckii and an endoparasite found on paper wasps have hundreds of photoreceptors in a single lens unlike conventional compound eyes. In particular, the eye structures of an adult Xenos peckii exhibit hundreds of photoreceptors on an individual eyelet and offer engineering inspiration for ultrathin cameras or imaging applications because they have higher visual acuity than other compound eyes. For instance, Xenos peckii’s eye-inspired cameras provide a 50 times higher spatial resolution than those based on arthropod eyes. In addition, the effective image resolution of the Xenos peckii’s eye can be further improved using the image overlaps between neighboring eyelets. This unique structure offers higher visual resolution than other insect eyes. The team achieved high-contrast and super-resolution imaging through a novel arrayed design of micro-optical elements comprising multilayered aperture arrays and inverted micro-lens arrays directly stacked over an image sensor. This optical component was integrated with a complementary metal oxide semiconductor image sensor. This is first demonstration of super-resolution imaging which acquires a single integrated image with high contrast and high resolving power reconstructed from high-contrast array images. It is expected that this ultrathin arrayed camera can be applied for further developing mobile devices, advanced surveillance vehicles, and endoscopes. Professor Jeong said, “This research has led to technological advances in imaging technology. We will continue to strive to make significant impacts on multidisciplinary research projects in the fields of microtechnology and nanotechnology, seeking inspiration from natural photonic structures.” This work was featured in Light Science & Applications last month and was supported by the National Research Foundation of Korea and the Ministry of Health and Welfare. < The ultrathin camera. > < Super-resolution imaging by array images. > Profile: Ki-Hun Jeong Professor kjeong@kaist.ac.kr http://biophotonics.kaist.ac.kr/ Department of Bio and Brain Engineering KAIST Profile: Kisoo Kim Ph.D. Candidate kisoo.kim1@kaist.ac.kr http://biophotonics.kaist.ac.kr/ Department of Bio and Brain Engineering KAIST
-
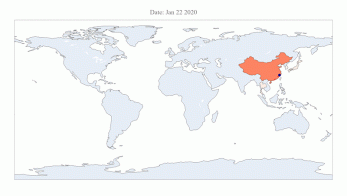
COVID-19 Map Shows How the Global Pandemic Spreads..
< COVID-19 Map > The COVID-19 map made by KAIST data scientists shows where and how the virus spreads from China, reportedly the epicenter of the disease. Professor Meeyoung Cha from the School of Computing and her group facilitated data based on the number of confirmed cases from January 22 to March 13 to analyze the trends of this global epidemic. The statistics include the number of confirmed cases, recoveries, and deaths across major continents based on the number of confirmed case data during that period. The moving dot on the map strikingly shows how the confirmed cases are moving across the globe. According to their statistics, the centroid of the disease starts from near Wuhan in China and moved to Korea, then through the European region via Italy and Iran. The data is collected by a graduate student, Sun Geng, who started the process during the time he was quarantined since coming back from his home in China. Gabriel Camilo Lima, an undergraduate student in the School of Computing who made the map, is now working remotely from his home in Brazil since all undergraduate students were required to move out of the dormitory last week. The university closed all undergraduate housing and advised the undergraduate students to go back home in a preventive measure to stop the virus from spreading across the campus. Gabriel said he calculated the centroid of all confirmed cases up to a given day. He explained, “I weighed each coordinate by the number of cases in that region and country and calculated an approximate center of gravity.” “The Earth is round, so the shortest path from Asia to Europe is often through Russia. In early March, the center of gravity of new cases was moving from Asia to Europe. Therefore, the centroid is moving to the west and goes through Russia, even though Russia has not reported many cases,” he said. Professor Cha, who is also responsible for the Data Science Group at the Institute for Basic Science as the Chief Investigator, said their group will continue to update the map using public data at https://ds.ibs.re.kr/index.php/covid-19/. < Sun Geng (Left) and Gabriel Camilo Lima (Right) >

